
Amide Nitroxide
Modern pulse EPR spectroscopy allows measurement of interspin distance distributions up to 8 nM. The precision of the measurement depends on the rigidity of the spin label with respect to the secondary structure examined.
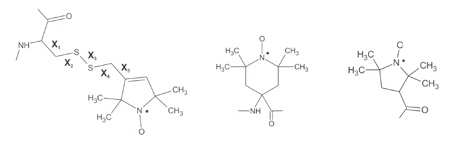
Nitroxide labels in use in biological systems (left - modified cysteine used in site-directed spin labeling; middle - semi-rigid TOAC fused to peptide backbone; right - proxylgroup, used to label amino terminals).
Novel amide-nitroxide spin labels have been developed to be incorporated into peptide backbones of proteins by expressed protein ligation.
More recently, DEER EPR spectroscopy was used to estimate the entropy of peptide binding to photoactivated rhodopsin using the TOAC and proxyl spin labels (Van Eps, N., Anderson, L.L., Kisselev, O.G., Baranski, T.J., Hubbell, W.L., and Marshall, G.R. 2010. EPR Studies of Functionally Active, Nitroxide Spin-Labeled Analogs of the C-Terminus of a G-Protein Alpha Subunit. Biochemistry in press).